Video by the National Science Foundation
Scrubbing bubbles
by Mason Adams
The collapse of a bubble triggers a black-hole effect on surrounding particles, pulling them toward the disappearing bubble.
The study of this bubble phenomenon began in an unlikely place: with boat propellers. Engineers found that cavitation—the process of bubble formation in fluids—damaged propellers run at higher speeds. The discovery led to related research on how cavitation might be used for medical and industrial applications, such as breaking up kidney stones or clumps of dirt in wastewater.
For a study published in the Physical Review Letters journal, Sunghwan "Sunny" Jung, an associate professor of biomedical engineering and mechanics, used a high-speed video camera to explore the effects of cavitation on objects.
Jung envisions that the collapsing bubbles' effect on particles may be used as a method for cleaning agricultural produce without chemical agents. He now is collaborating with researchers in the Department of Food Science and Technology to explore how small bubbles could draw microbes and dirt away from the skin of delicate produce, such as tomatoes and strawberries, without harming the crops.
How the physics of bursting bubbles may clean our food:
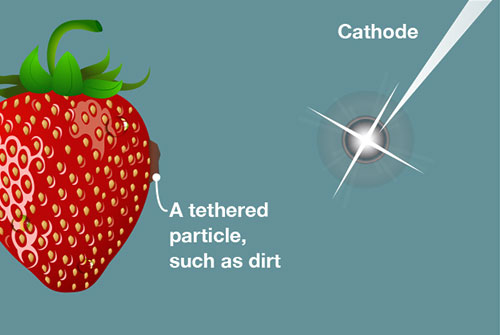
[1] An underwater spark increases the temperature.

[2] The increased temperature creates a bubble near a tethered particle.
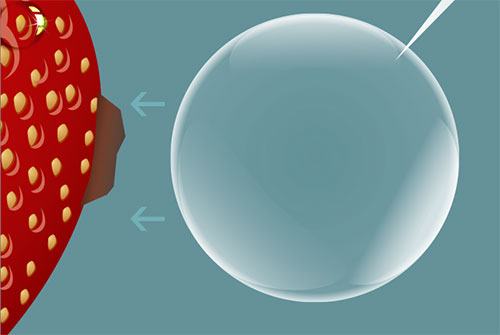
[3] The rapidly growing bubble begins to destabilize.

[4] As the bubble begins to collapse, a cone shape is created on the side facing the particle, due to a pressure differential.

[5] The bubble collapses as the temperature returns to normal.

[6] The pressure differential pulls the particle toward the space the bubble once occupied.